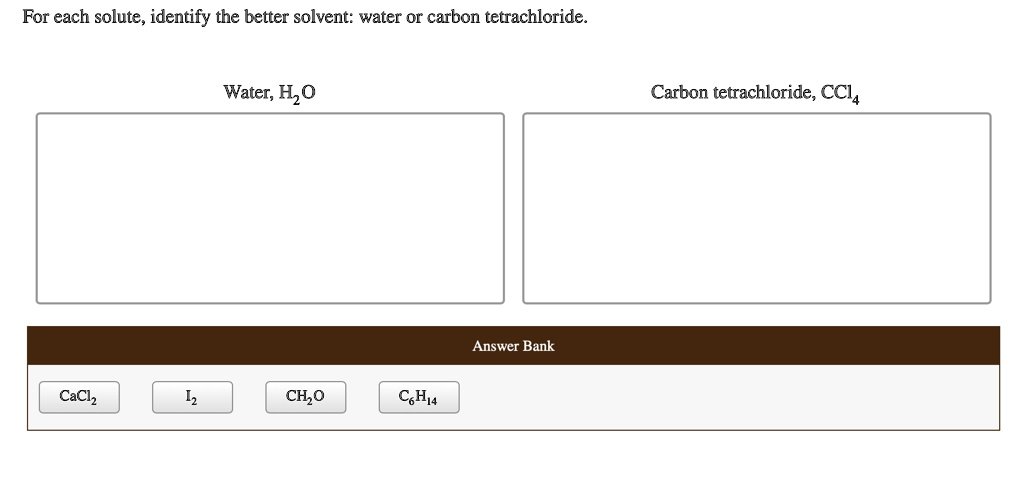
The first law of thermodynamics is concerned with the conservation of energy. It states that energy cannot be created or destroyed, only transferred or converted from one form to another. In mathematical terms, the change in internal energy of a system is equal to the heat added to the system minus the work done by the system.
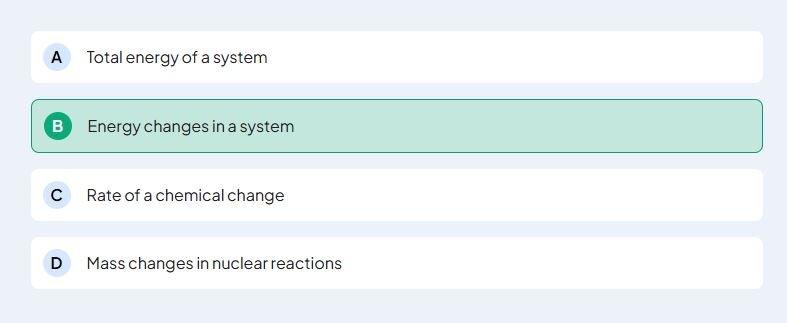
Text Solution – The first law of thermodynamics is concerned with the conservation of Energy
Here is how you can solve the question The first law of thermodynamics is concerned with the conservation of
To address the question about the first law of thermodynamics and its focus on conservation, we can follow these steps:
Step-by-Step Solution:
- Understanding the First Law of Thermodynamics:
- The first law of thermodynamics asserts that energy cannot be created or destroyed; it can only be converted from one form to another. This concept is referred to as the conservation of energy.
- Mathematical Expression:
- The first law can be written as:
- Where:
- ( Delta U ) = Change in the system’s internal energy
- ( Q ) = Heat added to the system
- ( W ) = Work done by the system
- Where:
- The first law can be written as:
- Identifying the Core Principle:
- From the equation, it’s clear that the change in internal energy (( Delta U )) depends on the heat added and the work done, emphasizing that energy is conserved within a closed system.
- Selecting the Correct Option:
- Among the options provided, we need to identify the one that reflects the conservation of energy. The correct choice will state that the first law of thermodynamics deals with energy conservation.
- Conclusion:
- Thus, the answer to the question is that the first law of thermodynamics focuses on the conservation of energy.
Final Answer:
- The first law of thermodynamics is concerned with the conservation of energy.